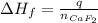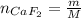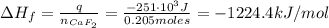Chemistry, 01.08.2020 23:01 kelsotay623
A 8.22-g sample of solid calcium reacted in excess fluorine gas to give a 16-g sample of pure solid CaF2. The heat given off in this reaction was 251 kJ at constant pressure. Given this information, what is the enthalpy of formation of CaF2(s)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2
You know the right answer?
A 8.22-g sample of solid calcium reacted in excess fluorine gas to give a 16-g sample of pure solid...
Questions


Mathematics, 27.01.2021 19:50

Mathematics, 27.01.2021 19:50

Mathematics, 27.01.2021 19:50


Chemistry, 27.01.2021 19:50

Computers and Technology, 27.01.2021 19:50

History, 27.01.2021 19:50


Mathematics, 27.01.2021 19:50

Mathematics, 27.01.2021 19:50



Biology, 27.01.2021 19:50


Mathematics, 27.01.2021 19:50

World Languages, 27.01.2021 19:50


Arts, 27.01.2021 19:50