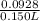Chemistry, 01.08.2020 23:01 alystidham1439
Suppose of nickel(II) iodide is dissolved in of a aqueous solution of potassium carbonate. Calculate the final molarity of nickel(II) cation in the solution. You can assume the volume of the solution doesn't change when the nickel(II) iodide is dissolved in it. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2


Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1

You know the right answer?
Suppose of nickel(II) iodide is dissolved in of a aqueous solution of potassium carbonate. Calculate...
Questions


English, 06.11.2020 07:30

History, 06.11.2020 07:30

English, 06.11.2020 07:30


Mathematics, 06.11.2020 07:30

History, 06.11.2020 07:30

Chemistry, 06.11.2020 07:30

Physics, 06.11.2020 07:30

Mathematics, 06.11.2020 07:30


Mathematics, 06.11.2020 07:30




Mathematics, 06.11.2020 07:30

Mathematics, 06.11.2020 07:30


Mathematics, 06.11.2020 07:30

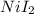 - 312.5 g
- 312.5 g