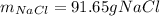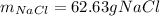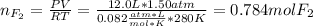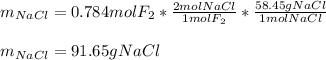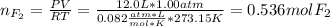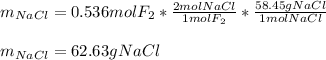Part 1. A chemist reacted 12.0 liters of F2 gas with NaCl in the laboratory to form Cl2 gas and NaF. Use the ideal gas law equation to determine the mass of NaCl that reacted with F2 at 280. K and 1.50 atm. F2 + 2NaCl → Cl2 + 2NaF Part 2. Explain how you would determine the mass of sodium chloride that can react with the same volume of fluorine gas at STP.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Part 1. A chemist reacted 12.0 liters of F2 gas with NaCl in the laboratory to form Cl2 gas and NaF....
Questions

English, 10.11.2020 07:30



Chemistry, 10.11.2020 07:30

Mathematics, 10.11.2020 07:30



Biology, 10.11.2020 07:30


World Languages, 10.11.2020 07:30

English, 10.11.2020 07:30




Mathematics, 10.11.2020 07:30

Chemistry, 10.11.2020 07:40

Mathematics, 10.11.2020 07:40

English, 10.11.2020 07:40


Chemistry, 10.11.2020 07:40