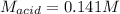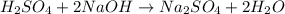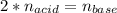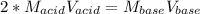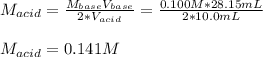Chemistry, 12.08.2020 05:01 KetaFord1978
g If the titration of a 10.0-mL sample of sulfuric acid requires 28.15 mL of 0.100 M sodium hydroxide, what is the molarity of the acid

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3


Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3

Chemistry, 23.06.2019 11:30
If 4.8 moles of x and 3.4 moles of y react according to the reaction below, how many moles of the excess reactant will be left over at the end of the reaction? 3x + 2y “yields”/ x3y2. a. 1.7 mol y left over b. 1.6 mol x left over c. 0.2 mol y left over d. 0.1 mol x left over
Answers: 1
You know the right answer?
g If the titration of a 10.0-mL sample of sulfuric acid requires 28.15 mL of 0.100 M sodium hydroxid...
Questions



History, 18.09.2019 09:00




Mathematics, 18.09.2019 09:00






Mathematics, 18.09.2019 09:00



Computers and Technology, 18.09.2019 09:00


Biology, 18.09.2019 09:00