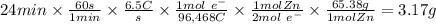Chemistry, 12.08.2020 06:01 sparkybig12
Zinc is used as a coating for steel to protect the steel from environmental corrosion. If a piece of steel is submerged in an electrolysis bath for 24 minutes with a current of 6.5 Amps, how many grams of zinc will be plated out? The molecular weight of Zn is 65.38, and Zn+2 + 2e– → Zn. Question 7 options: A) 3.17 g of Zn B) 1.09 g of Zn C) 6.34 g of Zn D) 12.68 g of Zn

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1


You know the right answer?
Zinc is used as a coating for steel to protect the steel from environmental corrosion. If a piece of...
Questions

History, 01.11.2019 21:31




English, 01.11.2019 21:31


Chemistry, 01.11.2019 21:31


Business, 01.11.2019 21:31



Mathematics, 01.11.2019 21:31

Biology, 01.11.2019 21:31


Mathematics, 01.11.2019 21:31