Chemistry, 12.08.2020 04:01 perezsamantha3oqr0za
H2S(g) 2H2O(l)3H2(g) SO2(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.60 moles of H2S(g) react at standard conditions.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3


Chemistry, 22.06.2019 15:00
Describe what happens to the molecules as water goes from ice to liquid to vapor. be sure to explain what happens to the temperature during the phase changes.
Answers: 2
You know the right answer?
H2S(g) 2H2O(l)3H2(g) SO2(g) Using standard absolute entropies at 298K, calculate the entropy change...
Questions




English, 30.03.2021 01:20


Mathematics, 30.03.2021 01:20



Mathematics, 30.03.2021 01:20


Physics, 30.03.2021 01:20



Mathematics, 30.03.2021 01:20



Mathematics, 30.03.2021 01:20



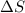 = 473.92J/K.mol
= 473.92J/K.mol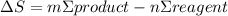
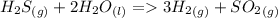
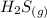 :
: