
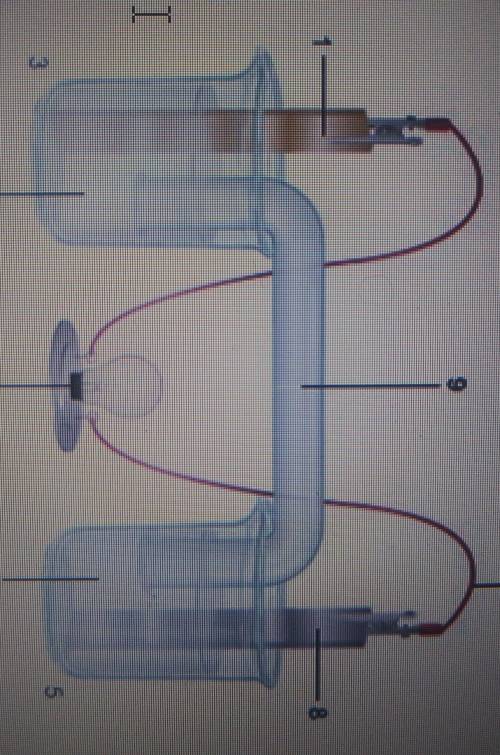
I need help asap. any help is appreciated.
the diagram shows an electrochemical cell with copper (left) and zinc (right) strips.
**Label all parts (1-9), including the solutions in each beaker and the connecting tube
**Label which cell is the cathode and which cell is the anode include the charge on each strip
**Show, or describe in detail, the flow of electrons
**Describe what type of electrochemical cell is pictured. Explain how the cell works Include the oxidation the oxidation half-reacions in your explanation

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 13:00
Imagine that you push on a large rock. at what point does your effort change the rock’s mechanical energy?
Answers: 1

Chemistry, 22.06.2019 19:30
How might this scientific phenomena be explained? a paper clip floats on water.
Answers: 1
You know the right answer?
I need help asap. any help is appreciated.
the diagram shows an electrochemical cell with copper (l...
Questions






Mathematics, 10.09.2021 18:40

Mathematics, 10.09.2021 18:40




Mathematics, 10.09.2021 18:40

English, 10.09.2021 18:40



Chemistry, 10.09.2021 18:40

History, 10.09.2021 18:40

Chemistry, 10.09.2021 18:40


Biology, 10.09.2021 18:40

Mathematics, 10.09.2021 18:40



