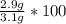Chemistry, 12.08.2020 07:01 kokokakahi
A student carries out the precipitation reaction shown below, starting with 0.030 moles of calcium nitrate. The final mass of the precipitate is 2.9 g. Answer the questions below to determine the percent yield. 3Ca(NO3)2(aq) + 2Na3PO4(aq) → Ca3(PO4)2(s) + 6NaNO3(aq) 1. a. Which product is the precipitate? b. How many moles of the precipitate would one expect to be produced from 0.030 moles of calcium nitrate? c. How many grams of solid do you expect to be produced? d. What is the percent yield?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1

You know the right answer?
A student carries out the precipitation reaction shown below, starting with 0.030 moles of calcium n...
Questions


History, 08.10.2019 18:30

Chemistry, 08.10.2019 18:30

Business, 08.10.2019 18:30

Health, 08.10.2019 18:30

Social Studies, 08.10.2019 18:30

Physics, 08.10.2019 18:30



SAT, 08.10.2019 18:30


Biology, 08.10.2019 18:30


English, 08.10.2019 18:30



Mathematics, 08.10.2019 18:30

Social Studies, 08.10.2019 18:30

Social Studies, 08.10.2019 18:30