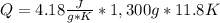Chemistry, 12.08.2020 08:01 runninglovexoxo
Calculate the energy required to heat 1.30kg of water from 22.4°C to 34.2°C . Assume the specific heat capacity of water under these conditions is 4.18·J·g−1K−1 . Round your answer to 3 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1
You know the right answer?
Calculate the energy required to heat 1.30kg of water from 22.4°C to 34.2°C . Assume the specific he...
Questions

History, 04.07.2019 22:40



Mathematics, 04.07.2019 22:40


English, 04.07.2019 22:40

Advanced Placement (AP), 04.07.2019 22:40


History, 04.07.2019 22:40

Business, 04.07.2019 22:40

Biology, 04.07.2019 22:40





Mathematics, 04.07.2019 22:40

History, 04.07.2019 22:40



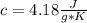 m= 1.30 kg= 1,300 g (1 kg=1,000 g)ΔT= 34.2 °C - 22.4 °C= 11.8 °C= 11.8 °K Being a temperature difference, it is independent if they are degrees Celsius or degrees Kelvin. That is, the temperature difference is the same in degrees Celsius or degrees Kelvin.
m= 1.30 kg= 1,300 g (1 kg=1,000 g)ΔT= 34.2 °C - 22.4 °C= 11.8 °C= 11.8 °K Being a temperature difference, it is independent if they are degrees Celsius or degrees Kelvin. That is, the temperature difference is the same in degrees Celsius or degrees Kelvin.