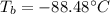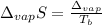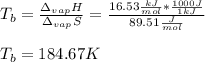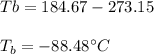Chemistry, 14.08.2020 01:01 jacobdesalvo9890
The ΔHvap of nitrous oxide is 16.53 kJ · mol−1 and its ΔSvap is 89.51 J · mol−1 · K−1. What it the boiling point of nitrous oxide?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1
You know the right answer?
The ΔHvap of nitrous oxide is 16.53 kJ · mol−1 and its ΔSvap is 89.51 J · mol−1 · K−1. What it the b...
Questions


Business, 11.10.2019 09:30

Mathematics, 11.10.2019 09:30

Mathematics, 11.10.2019 09:30



Mathematics, 11.10.2019 09:30


English, 11.10.2019 09:30



Mathematics, 11.10.2019 09:30

History, 11.10.2019 09:30

Mathematics, 11.10.2019 09:30



Computers and Technology, 11.10.2019 09:30


Mathematics, 11.10.2019 09:30