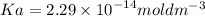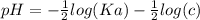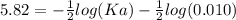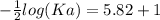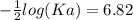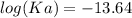Chemistry, 13.08.2020 17:01 lucifer6669
The pH of an acid solution is 5.82. Calculate the Ka for the monoprotic acid. The initial acid concentration is 0.010 M.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 00:30
What does x represent in the formula for the compound xcl4?
Answers: 1

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
You know the right answer?
The pH of an acid solution is 5.82. Calculate the Ka for the monoprotic acid. The initial acid conce...
Questions



History, 05.05.2020 01:46


Physics, 05.05.2020 01:46

Physics, 05.05.2020 01:46



Physics, 05.05.2020 01:46




Mathematics, 05.05.2020 01:46


Chemistry, 05.05.2020 01:46

Mathematics, 05.05.2020 01:46



English, 05.05.2020 01:46

English, 05.05.2020 01:46