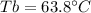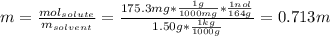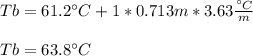Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 23.06.2019 01:00
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
You know the right answer?
A 175.3 mg sample of C10H12O2 was dissolved in 1.50 g of chloroform (Kb = 3.63 °C/m). What is the bo...
Questions

English, 07.10.2021 16:50



Social Studies, 07.10.2021 16:50



Social Studies, 07.10.2021 16:50







Mathematics, 07.10.2021 16:50

Business, 07.10.2021 16:50

Computers and Technology, 07.10.2021 16:50


Law, 07.10.2021 16:50

Business, 07.10.2021 16:50