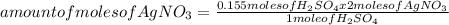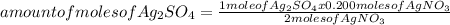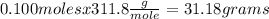Chemistry, 15.08.2020 23:01 cesarcastellan9
If 0.200 moles of AgNO₃ react with 0.155 moles of H₂SO₄ according to this UNBALANCED equation below, what is the mass in grams of Ag₂SO₄ that could be formed? AgNO₃(aq) + H₂SO₄ (aq) → Ag₂SO₄ (s) + HNO₃ (aq)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1


Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
If 0.200 moles of AgNO₃ react with 0.155 moles of H₂SO₄ according to this UNBALANCED equation below,...
Questions

Mathematics, 12.02.2021 02:50

Chemistry, 12.02.2021 02:50

Mathematics, 12.02.2021 02:50

Mathematics, 12.02.2021 02:50


Mathematics, 12.02.2021 02:50



English, 12.02.2021 02:50

Physics, 12.02.2021 02:50

Chemistry, 12.02.2021 02:50

Mathematics, 12.02.2021 02:50

Mathematics, 12.02.2021 02:50





Biology, 12.02.2021 02:50

Mathematics, 12.02.2021 02:50