
Chemistry, 18.08.2020 17:01 cluchmasters5634
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reaction mixture have on the system?
CuS(s) + O2(g) ? Cu(s) + SO2(g)
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reaction mixture have on the system?
CuS(s) + O2(g) ? Cu(s) + SO2(g)
a) The equilibrium constant will increase.
b) The reaction will shift to the left in the direction of reactants.
c) The reaction will shift to the right in the direction of products.
d) No effect will be observed.
e) The equilibrium constant will decrease.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reac...
Questions



Mathematics, 29.08.2020 22:01








Computers and Technology, 29.08.2020 22:01


Business, 29.08.2020 22:01

English, 29.08.2020 22:01

Mathematics, 29.08.2020 22:01




Social Studies, 29.08.2020 22:01

![Q = \displaystyle \frac{[\mathrm{SO_2\, (g)}]}{[\mathrm{O_2\, (g)}]}](/tpl/images/0723/8610/a7d3d.png) .
.![[\mathrm{SO_2\, (g)}]](/tpl/images/0723/8610/2691f.png) and
and ![[\mathrm{O_2\, (g)}]](/tpl/images/0723/8610/46794.png) will increase if the pressure is increased through compression. However, because
will increase if the pressure is increased through compression. However, because 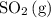 and
and 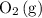 have the same coefficients in the equation, their concentrations are raised to the same power in the equilibrium quotient
have the same coefficients in the equation, their concentrations are raised to the same power in the equilibrium quotient 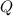 .
. 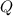 . If the system was already at equilibrium, it will continue to be at an equilibrium even after the change to its pressure. Therefore, no overall effect on the equilibrium position should be visible.
. If the system was already at equilibrium, it will continue to be at an equilibrium even after the change to its pressure. Therefore, no overall effect on the equilibrium position should be visible.

