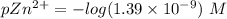Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Select each correct answer. more than one answer may be correct. which of the following is a characteristic of unicellular organisms? they can possess tissues and organs. all of their functions are performed by a single cell. they are usually microscopic. each of their cells is specialized to perform a specific function.
Answers: 1


Chemistry, 22.06.2019 12:00
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1
You know the right answer?
Calculate the pZn of a solution prepared by mixing 25.0 mL of 0.0100 M EDTA with 50.0 mL of 0.00500...
Questions

Mathematics, 30.11.2019 00:31




English, 30.11.2019 00:31


Biology, 30.11.2019 00:31




Mathematics, 30.11.2019 00:31


History, 30.11.2019 00:31


History, 30.11.2019 00:31


Biology, 30.11.2019 00:31


History, 30.11.2019 00:31

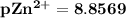
![pH = pKa[NH^+_4] + log \dfrac{[NH_3]}{[NH_4^+]}](/tpl/images/0726/1828/94d35.png)
 = 9.26
= 9.26 = 0.100 M
= 0.100 M 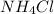 = 0.176 M
= 0.176 M![pH = 9.26 + log \dfrac{[0.100]}{[0.176]}](/tpl/images/0726/1828/11e21.png)
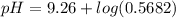
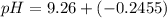
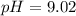
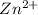 and EDTA is :
and EDTA is :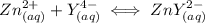
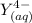 denotes the fully deprotonated form of the EDTA
denotes the fully deprotonated form of the EDTA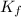 of the equation for the reaction can be represented as:
of the equation for the reaction can be represented as:![K_f = \dfrac{[ZnY^{2-}]}{[Zn^{2+} ][Y^{4-}]}](/tpl/images/0726/1828/c7b1d.png) ----- (1)
----- (1)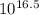
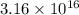
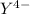 ,
,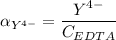
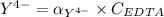
![K_f = \dfrac{[ZnY^{2-}]}{[Zn^{2+} ] \ \ \alpha_ {Y^{4-}} \times {C_{EDTA}}}](/tpl/images/0726/1828/75df9.png)
![K_f' = K_f \times \alpha _Y{^4-} = \dfrac{[ZnY^{2-}]}{[Zn^{2+} ] \ C_{EDTA} }](/tpl/images/0726/1828/a3102.png)
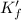 = conditional formation constant
= conditional formation constant 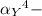 = the fraction of EDTA that exit in the form of the presences of the 4 charges .
= the fraction of EDTA that exit in the form of the presences of the 4 charges .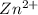 initially in titrand is now present in
initially in titrand is now present in 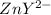
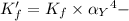
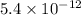
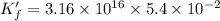
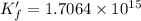
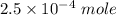
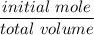
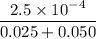
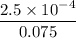
![K_f' = \dfrac{[ZnY^{2-}]}{[Zn^{2+} ] \ C_{EDTA} }](/tpl/images/0726/1828/c89c2.png)
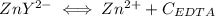
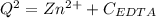
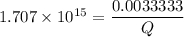
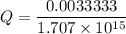
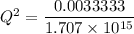
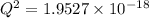
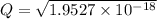
 M
M![[Zn^{2+}]= 1.39 \times 10^{-9} \ M](/tpl/images/0726/1828/63850.png)
![pZn ^{2+} =- log [Zn^{2+}]](/tpl/images/0726/1828/3ecce.png)