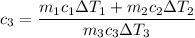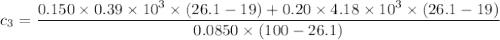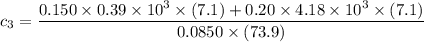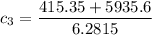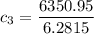Chemistry, 22.08.2020 01:01 Ramone7415
A laboratory technician drops a 0.0850 kg sample of unknown solid material, at a temperature of 100 oC, into a calorimeter. The calorimeter can, initially at 19.0 oC, is made of 0.150 kg of copper and contains 0.20 kg of water. The final temperature of the calorimeter can, and contents is 26.1 oC. Compute the specific heat of the sample.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1

Chemistry, 23.06.2019 05:30
What is the morality of 2.50 l of solution that contains 1.85 mol of anhydrous sodium tetraborate?
Answers: 1
You know the right answer?
A laboratory technician drops a 0.0850 kg sample of unknown solid material, at a temperature of 100...
Questions

Mathematics, 02.03.2021 18:40

Social Studies, 02.03.2021 18:40


History, 02.03.2021 18:40

Chemistry, 02.03.2021 18:40

Biology, 02.03.2021 18:40


Mathematics, 02.03.2021 18:40

Geography, 02.03.2021 18:40

Mathematics, 02.03.2021 18:40


Mathematics, 02.03.2021 18:40


Mathematics, 02.03.2021 18:40


Social Studies, 02.03.2021 18:40


English, 02.03.2021 18:40


Biology, 02.03.2021 18:40

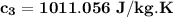
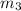 = 0.0850
= 0.0850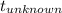 = 100° C
= 100° C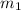 = 0.150 kg
= 0.150 kg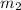 = 0.20 kg
= 0.20 kg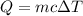
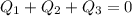
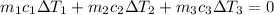
 can be computed by making
can be computed by making