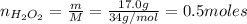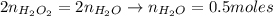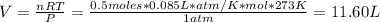Chemistry, 27.08.2020 04:01 kmshacklette9879
Hydrogen peroxide (H2O2, 34 g/mol) decomposes into water vapor and oxygen gas. How many liters of water vapor are produced from the decomposition of 17.0 g of H2O2 at STP?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3

Chemistry, 22.06.2019 23:00
What is the formula of the ionic compound composed of calcium cations and chloride anions
Answers: 1

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
Hydrogen peroxide (H2O2, 34 g/mol) decomposes into water vapor and oxygen gas. How many liters of wa...
Questions


Mathematics, 25.11.2020 04:30



Medicine, 25.11.2020 04:30

Health, 25.11.2020 04:30

English, 25.11.2020 04:30

Biology, 25.11.2020 04:30


Mathematics, 25.11.2020 04:30

Mathematics, 25.11.2020 04:30


Mathematics, 25.11.2020 04:30

Biology, 25.11.2020 04:30


Mathematics, 25.11.2020 04:30


Chemistry, 25.11.2020 04:30

Mathematics, 25.11.2020 04:30