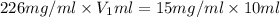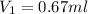Chemistry, 27.08.2020 23:01 joelpimentel
You have a stock solution that is 226mg/mL and you need 10mL of a working solution that is 15mg/mL. What is the volume of stock solution will you need to dilute to have 10mL of working solution at the above concentration. State your answer to two decimals.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 23.06.2019 04:31
Pls i will do pls imma diewhat forms white light? (4 points)a. combination of all wavelengths of ultraviolet light b. combination of all wavelengths of visible lightc. absorption of electromagnetic waves d. absorption of infrared rays
Answers: 2

Chemistry, 23.06.2019 10:10
In a covalent bond, two atoms are held together by the attraction between . the number of covalent bonds that an atom can form depends on the number of in the atom.
Answers: 2
You know the right answer?
You have a stock solution that is 226mg/mL and you need 10mL of a working solution that is 15mg/mL....
Questions

Computers and Technology, 26.02.2020 03:18


Mathematics, 26.02.2020 03:18




History, 26.02.2020 03:18


Chemistry, 26.02.2020 03:18

Mathematics, 26.02.2020 03:18


Mathematics, 26.02.2020 03:18



Computers and Technology, 26.02.2020 03:19

Mathematics, 26.02.2020 03:19




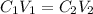
 = concentration of stock solution = 226 mg/ml
= concentration of stock solution = 226 mg/ml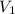 = volume of stock solution = ?
= volume of stock solution = ? = concentration of working solution= 15 mg/ml
= concentration of working solution= 15 mg/ml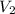 = volume of working solution= 10 ml
= volume of working solution= 10 ml