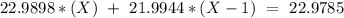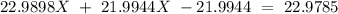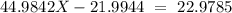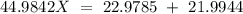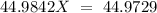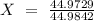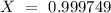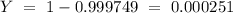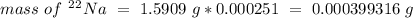Chemistry, 31.08.2020 01:01 Blackhawk1881
Sodium only has one naturally occuring isotope, Na23 , with a relative atomic mass of 22.9898 u. A synthetic, radioactive isotope of sodium, Na22 , is used in positron emission tomography. Na22 has a relative atomic mass of 21.9944 u. A 1.5909 g sample of sodium containing a mixture of Na23 and Na22 has an apparent "atomic mass" of 22.9785 u . Find the mass of Na22 contained in this sample

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Much of the general structure and physical properties of the interior of the earth are inferred from: a)deep oil and gas bore holes b)geologic investigations c)analysis of seismic waves d) study of volcanoes
Answers: 1

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1
You know the right answer?
Sodium only has one naturally occuring isotope, Na23 , with a relative atomic mass of 22.9898 u. A s...
Questions

Mathematics, 21.01.2021 20:30

Mathematics, 21.01.2021 20:30

English, 21.01.2021 20:30

Social Studies, 21.01.2021 20:30


Mathematics, 21.01.2021 20:30



Computers and Technology, 21.01.2021 20:30

Mathematics, 21.01.2021 20:30

Mathematics, 21.01.2021 20:30



Mathematics, 21.01.2021 20:30

History, 21.01.2021 20:30


Social Studies, 21.01.2021 20:30



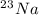 is an unknow value "X" and the molar fraction of
is an unknow value "X" and the molar fraction of 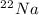 is "Y". We have to keep in mind that the molar fractions can be added:
is "Y". We have to keep in mind that the molar fractions can be added: