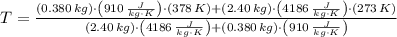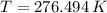Chemistry, 01.09.2020 02:01 davidcortez27
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then placed in 2.40 kg of water that is at 293 K. If the system is left to reach its equilibrium state, what will the equilibrium temperature be? Assume that no thermal energy is lost to the external environment. Use 4.186 J/(g x K) as the specific heat of water.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1
You know the right answer?
A 0.380 kg sample of aluminum (with a specific heat of 910.0 J/(kg x K)) is heated to 378 K and then...
Questions

Mathematics, 27.08.2019 13:30

English, 27.08.2019 13:30

Mathematics, 27.08.2019 13:30

Mathematics, 27.08.2019 13:30

Mathematics, 27.08.2019 13:30


Mathematics, 27.08.2019 13:30


Chemistry, 27.08.2019 13:30


Biology, 27.08.2019 13:30

Mathematics, 27.08.2019 13:30




Physics, 27.08.2019 13:30

English, 27.08.2019 13:30

Mathematics, 27.08.2019 13:30


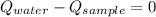
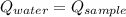
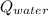 - Heat received by water, measured in joules.
- Heat received by water, measured in joules.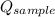 - Heat released by the sample of aluminium, measured in joules.
- Heat released by the sample of aluminium, measured in joules.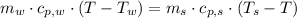
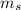 ,
, 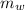 - Mass of water and the sample of aluminium, measured in kilograms.
- Mass of water and the sample of aluminium, measured in kilograms.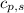 ,
, 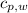 - Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.
- Specific heats of the sample of aluminium and water, measured in joules per kilogram-Kelvin.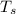 ,
, 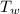 - Initial temperatures of the sample of aluminium and water, measured in Kelvin.
- Initial temperatures of the sample of aluminium and water, measured in Kelvin.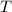 - Temperature which system reaches thermal equilibrium, measured in Kelvin.
- Temperature which system reaches thermal equilibrium, measured in Kelvin.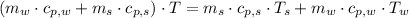
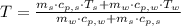
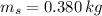 ,
, 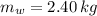 ,
, 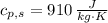 ,
, 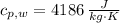 ,
, 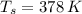 and
and 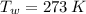 , the final temperature of the system is:
, the final temperature of the system is: