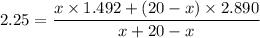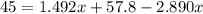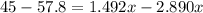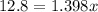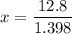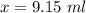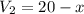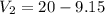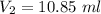Suppose now that you wanted to determine the density of a small crystal to confirm that it is graphite. From the literature, you know that graphite has a density of 2.25 g/cm^3. How would you prepare 20.0 mL of the liquid mixture having that density from pure samples of CHCl3 (d = 1.492 g/mL) and CHBr3 (d = 2.890 g/mL)? (Note: 1 mL = 1 cm^3.)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:30
This question is about electrolysis. metal spoons can be coated with silver. this is called electroplating. suggest one reason why spoons are electroplated?
Answers: 1

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
Suppose now that you wanted to determine the density of a small crystal to confirm that it is graphi...
Questions

Mathematics, 03.09.2021 07:00

Biology, 03.09.2021 07:00


Mathematics, 03.09.2021 07:00


Computers and Technology, 03.09.2021 07:00


Spanish, 03.09.2021 07:00


Mathematics, 03.09.2021 07:00

Mathematics, 03.09.2021 07:00

Mathematics, 03.09.2021 07:00


Chemistry, 03.09.2021 07:00

Biology, 03.09.2021 07:00



Mathematics, 03.09.2021 07:00


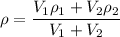
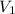 = volume of first compound
= volume of first compound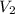 = volume of second compound
= volume of second compound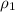 =density of first compound
=density of first compound