Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 06:30
1.17 mol hcl and 2.5 mol naoh react according to the equation hcl + naoh -> nacl + h2o . if the limiting reactant is hcl, determine the amount of excess reactant that remains. answer in units of mol.
Answers: 1
You know the right answer?
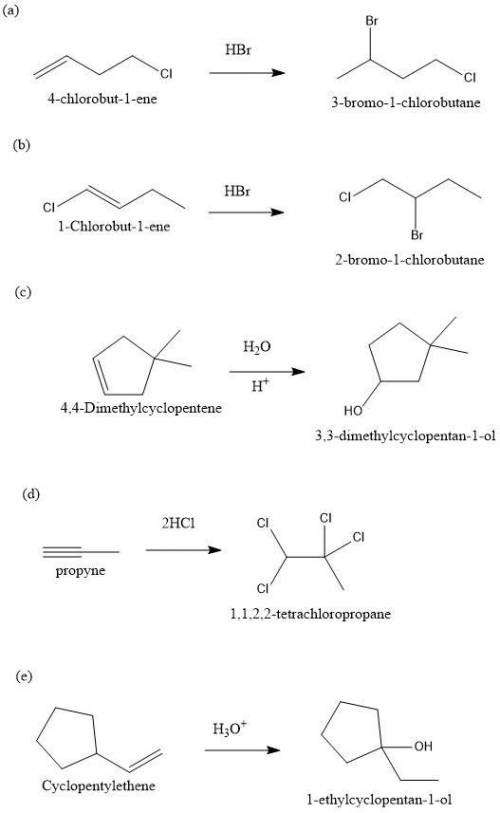
Predict the major product(s) for each of the following reactions. H3O (a) 4-Chlorobut-1-ene HBr (b)...
Questions


Business, 22.09.2021 14:00

English, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00





Chemistry, 22.09.2021 14:00

Chemistry, 22.09.2021 14:00

Mathematics, 22.09.2021 14:00

Chemistry, 22.09.2021 14:00


English, 22.09.2021 14:00


Mathematics, 22.09.2021 14:00


Mathematics, 22.09.2021 14:00

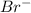 " will be added to the most substituted carbon. In this case carbon 3 to produce 3-bromo-1-chlorobutane.
" will be added to the most substituted carbon. In this case carbon 3 to produce 3-bromo-1-chlorobutane.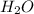 " will be added to the most substituted carbon. In this case carbon 3 to produce 3,3-dimethylcyclopentan-1-ol.
" will be added to the most substituted carbon. In this case carbon 3 to produce 3,3-dimethylcyclopentan-1-ol.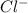 " will be added to the most substituted carbon, but we have 2 moles of the nucleophile, so would be added 2 times and we will have as product 1,1,2,2-tetrachloropropane.
" will be added to the most substituted carbon, but we have 2 moles of the nucleophile, so would be added 2 times and we will have as product 1,1,2,2-tetrachloropropane.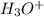 " will be added to the most substituted carbon. But in this case, the carbon cation would be produced in carbon 1 of the ethene. So, we can have a hydride shift to produce a tertiary carbocation. With this in mind the nucleohile will be addde to this tertiary carbocation and we will have 1-ethylcyclopentan-1-ol.
" will be added to the most substituted carbon. But in this case, the carbon cation would be produced in carbon 1 of the ethene. So, we can have a hydride shift to produce a tertiary carbocation. With this in mind the nucleohile will be addde to this tertiary carbocation and we will have 1-ethylcyclopentan-1-ol.