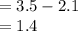Which of the following molecules contains polar covalent bonds?
O H2
O H20
O NaCl
...


Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1
You know the right answer?
Questions

Mathematics, 15.01.2021 09:30


Spanish, 15.01.2021 09:30

Arts, 15.01.2021 09:30

Chemistry, 15.01.2021 09:30

Mathematics, 15.01.2021 09:30


Mathematics, 15.01.2021 09:30





Biology, 15.01.2021 09:30

History, 15.01.2021 09:30

Biology, 15.01.2021 09:30

Mathematics, 15.01.2021 09:30

Mathematics, 15.01.2021 09:30

History, 15.01.2021 09:30

Social Studies, 15.01.2021 09:30

Mathematics, 15.01.2021 09:30