
Chemistry, 05.09.2020 02:01 dieulynx1171
What is the freezing point (°C) of a solution prepared by dissolving 11.3 g of Ca(NO3)2 (formula weight = 164 g/mol) in 115 g of water? The molal freezing point depression constant for water is 1.86 °C/m.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Describe the chemical reaction based on the chemical equation below. also, explain whether the equation is balanced.
Answers: 1



Chemistry, 22.06.2019 18:50
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
You know the right answer?
What is the freezing point (°C) of a solution prepared by dissolving 11.3 g of Ca(NO3)2 (formula wei...
Questions



Biology, 09.04.2020 05:06





Business, 09.04.2020 05:07

English, 09.04.2020 05:07







English, 09.04.2020 05:08




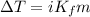
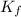 = 1.86 °C/m
= 1.86 °C/m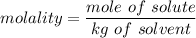
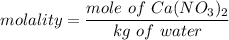
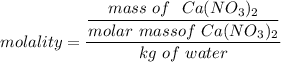
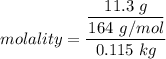
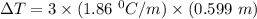
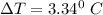
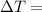 the freezing point of water - freezing point of the solution
the freezing point of water - freezing point of the solution

