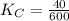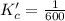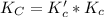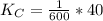Chemistry, 06.09.2020 02:01 Deavionaaaaa
Given the equilibrium constants for the following two reactions at a 298K:NiO(s) + H2(g) ⇌ Ni(s) + H2O(g) Kc=40NiO(s) +CO(g) ⇌ Ni(s) +CO2(g) Kc=600Calculate the value for the equilibrium constant, Kc, for the reaction:CO2(g) + H2(g) ⇌ CO(g) + H2O(g)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which of the following statements is true about planck’s law
Answers: 1

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 08:40
What is the value of keq for the reaction expressed in scientific notation?
Answers: 1

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1
You know the right answer?
Given the equilibrium constants for the following two reactions at a 298K:NiO(s) + H2(g) ⇌ Ni(s) + H...
Questions

Biology, 05.07.2019 10:30

Social Studies, 05.07.2019 10:30

History, 05.07.2019 10:30

Social Studies, 05.07.2019 10:30




French, 05.07.2019 10:30


History, 05.07.2019 10:30


Mathematics, 05.07.2019 10:30


History, 05.07.2019 10:30


Mathematics, 05.07.2019 10:30


Social Studies, 05.07.2019 10:30

Spanish, 05.07.2019 10:30