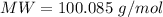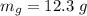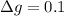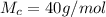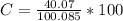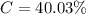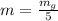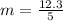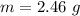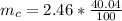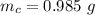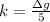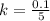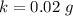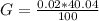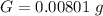Chemistry, 10.09.2020 03:01 kragland4752
g Five calcite, CaCO3 (MW 100.085 g/mol), samples of equal mass have a total mass of 12.3±0.1 g. What is the absolute uncertainty (grams) of calcium in each average calcium mass of the sample? Assume that the relative uncertainties in atomic mass are small compared the uncertainty of the total mass.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1

Chemistry, 23.06.2019 06:30
Achemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature: 2 no(g) + cl2(g) < => 2 nocl(g) kp = 2 x 10^(-6)he fills a reaction vessel at this temperature with 13. atm of nitrogen monoxide gas and 12. atm of chlorine gas. use this data to answer the questions: a. can you predict the equilibrium pressure of noci, using only the tools available to you within aleks? y/nb. if you said yes, then enter the equilibrium pressure of nocl at right. round your answer to 1 significant digit.
Answers: 1
You know the right answer?
g Five calcite, CaCO3 (MW 100.085 g/mol), samples of equal mass have a total mass of 12.3±0.1 g. Wha...
Questions


World Languages, 18.11.2020 03:00

English, 18.11.2020 03:00

History, 18.11.2020 03:00


Mathematics, 18.11.2020 03:00

Mathematics, 18.11.2020 03:00



English, 18.11.2020 03:00


Advanced Placement (AP), 18.11.2020 03:00

Mathematics, 18.11.2020 03:00



English, 18.11.2020 03:00

English, 18.11.2020 03:00

English, 18.11.2020 03:00


Mathematics, 18.11.2020 03:00

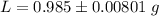
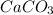 is
is