
Chemistry, 10.09.2020 03:01 CoreyHammond1517
When 229.0 J of energy is supplied as heat to 3.00 mol of Ar(g) at constant pressure the temperature of the sample increases by 2.55 K. Assuming that in the experiment the gas behaves as an ideal gas, calculate the molar heat capacities at constant volume and at constant pressure of Ar(g).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 22:00
Imagine one batch of soup (batch “a”) is made with 8.19 g/can of salt, according to the recipe, and a second batch of soup (batch “b”) is made with 8.32 g/can of salt. explain which batch would be more resistant to frost damage if it is shipped a great distance in winter and explain why.
Answers: 2
You know the right answer?
When 229.0 J of energy is supplied as heat to 3.00 mol of Ar(g) at constant pressure the temperature...
Questions

Mathematics, 22.04.2021 01:20



Mathematics, 22.04.2021 01:20



English, 22.04.2021 01:20



English, 22.04.2021 01:20

Chemistry, 22.04.2021 01:20

Mathematics, 22.04.2021 01:20

English, 22.04.2021 01:20

Mathematics, 22.04.2021 01:20






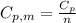
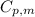 is the molar heat capacity at constant pressure
is the molar heat capacity at constant pressure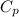 is the heat capacity at constant pressure
is the heat capacity at constant pressure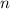 is the number of moles
is the number of moles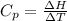
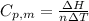
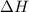 = 229.0 J
= 229.0 J = 2.55 K
= 2.55 K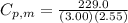
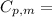 29.93 JK⁻¹mol⁻¹
29.93 JK⁻¹mol⁻¹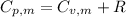
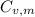 is the molar heat capacity at constant volume
is the molar heat capacity at constant volume 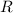 is the gas constant (
is the gas constant (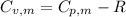
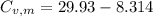
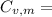 21.62 JK⁻¹mol⁻¹
21.62 JK⁻¹mol⁻¹

