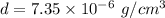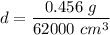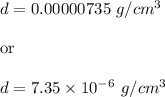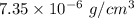Chemistry, 20.09.2020 15:01 Vishal123180
What is the density of an object with a volume of 62 L and a mass of 456 mg?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
What are the major products produced in the combustion of c10h22 under the following conditions? write balanced chemical equations for each. a. an excess of oxygen b. a slightly limited oxygen supply c. a very limited supply of oxygen d. the compound is burned in air
Answers: 2

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2
You know the right answer?
What is the density of an object with a volume of 62 L and a mass of 456 mg?...
Questions


Mathematics, 10.12.2020 22:30

English, 10.12.2020 22:30

History, 10.12.2020 22:30

Mathematics, 10.12.2020 22:30

Mathematics, 10.12.2020 22:30

World Languages, 10.12.2020 22:30

Mathematics, 10.12.2020 22:30


Social Studies, 10.12.2020 22:30


Mathematics, 10.12.2020 22:30

Mathematics, 10.12.2020 22:30

Mathematics, 10.12.2020 22:30

Biology, 10.12.2020 22:30


English, 10.12.2020 22:30

Chemistry, 10.12.2020 22:30

Spanish, 10.12.2020 22:30