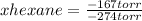A solution contains a mixture of pentane and hexane at room temperature. The solution has a vapor pressure of 258 torr . Pure pentane and hexane have vapor pressures of 425 torr and 151 torr, respectively, at room temperature. What is the mole fraction of hexane? (Assume ideal behavior.)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 23.06.2019 00:00
The graph indicates the running route for tobias. which best describes his run? from time 0 to 6, he went fast and then slowed down. from time 6 to 10, he was at his slowest. from time 12 to 14, he went very slow. from time 14 to 18, he went toward the starting point.
Answers: 2

Chemistry, 23.06.2019 04:40
[01.07]what is the answer to the problem: 101 g + 25.01 g + 5.05 g? 131.06 g 131.1 g 131 g 130 g
Answers: 1

You know the right answer?
A solution contains a mixture of pentane and hexane at room temperature. The solution has a vapor pr...
Questions

Mathematics, 30.07.2019 15:30



History, 30.07.2019 15:30


Chemistry, 30.07.2019 15:30









Spanish, 30.07.2019 15:30

Physics, 30.07.2019 15:30

Biology, 30.07.2019 15:30