
Chemistry, 21.09.2020 14:01 cassmoney94
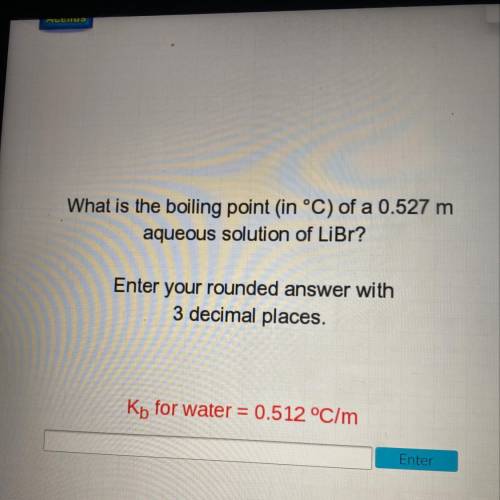
What is the boiling point (in °C) of a 0.527 m
aqueous solution of LiBr?
Enter your rounded answer with
3 decimal places.
Kb for water = 0.512 °C/m

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
What is the boiling point (in °C) of a 0.527 m
aqueous solution of LiBr?
Enter your rounded a...
Enter your rounded a...
Questions

Mathematics, 29.04.2021 01:00

Mathematics, 29.04.2021 01:00


Mathematics, 29.04.2021 01:00

Biology, 29.04.2021 01:00

Mathematics, 29.04.2021 01:00


Mathematics, 29.04.2021 01:00



History, 29.04.2021 01:00


Mathematics, 29.04.2021 01:00


Chemistry, 29.04.2021 01:00

Health, 29.04.2021 01:00







