5 points


Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2
You know the right answer?
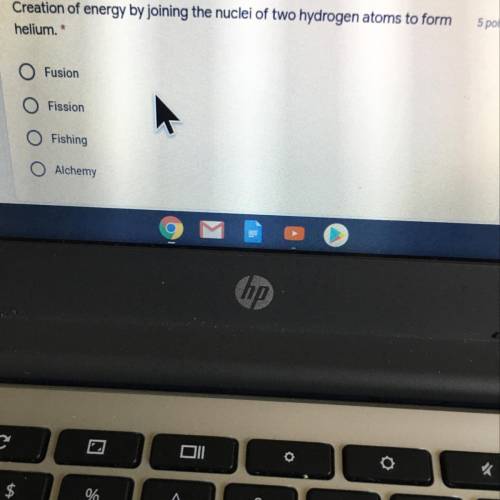
Creation of energy by joining the nuclei of two hydrogen atoms to form
helium. *
5 points
5 points
Questions


English, 09.03.2021 19:40

Mathematics, 09.03.2021 19:40



Mathematics, 09.03.2021 19:40


Mathematics, 09.03.2021 19:40



Mathematics, 09.03.2021 19:40





Mathematics, 09.03.2021 19:40


Medicine, 09.03.2021 19:40

Mathematics, 09.03.2021 19:40