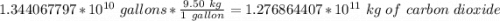Be sure to answer all parts. One gallon of gasoline in an automobiles engine produces on average 9.50 kg of carbon dioxide, which is a greenhouse gas; that is, it promotes the warming of Earth's atmosphere. Calculate the annual production of carbon dioxide in kilograms if there are exactly 40.0 million cars in the United States and each car covers a distance of 7930 mi at a consumption rate of 23.6 miles per gallon. Enter your answer in scientific notation.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1

Chemistry, 23.06.2019 06:30
Which of these describes how heat is transferred by convection* a. sunlight travels through space without the aid of fluids or solids. b. warm air rises and takes the heat with it, eventually, it cools and sinks c. air at the equator rises and sinks at the poles. d. air molecules touch the warm ground, heating them up *not conduction
Answers: 3

Chemistry, 23.06.2019 12:00
In ccl4 is there a certain length that the main atom carbon is a certain distance from the chlorine’s?
Answers: 1
You know the right answer?
Be sure to answer all parts. One gallon of gasoline in an automobiles engine produces on average 9.5...
Questions



Mathematics, 21.05.2021 09:50

Mathematics, 21.05.2021 09:50



Mathematics, 21.05.2021 09:50

History, 21.05.2021 09:50

Biology, 21.05.2021 14:00

History, 21.05.2021 14:00

Biology, 21.05.2021 14:00








English, 21.05.2021 14:00