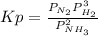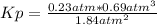Chemistry, 24.09.2020 07:01 20jessicacabriales
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studying this reaction fills a flask with of ammonia gas, and when the mixture has come to equilibrium measures the partial pressure of hydrogen gas to be . Calculate the pressure equilibrium constant for the decomposition of ammonia at the final temperature of the mixture. Round your answer to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1
You know the right answer?
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studyin...
Questions


Mathematics, 18.03.2021 22:20



Physics, 18.03.2021 22:20



Mathematics, 18.03.2021 22:20

Advanced Placement (AP), 18.03.2021 22:20

Mathematics, 18.03.2021 22:20



Mathematics, 18.03.2021 22:20





Mathematics, 18.03.2021 22:20

Mathematics, 18.03.2021 22:20