
Chemistry, 29.09.2020 23:01 COOLIOMARIS
Using the enthalpy diagram below, determine which of the following is true.
1. The reaction is exothermic
2. The energy difference between reactants and products is greater for the reverse direction than for the forward direction.
3. The combined reactants possess more energy than the combined products.
4. The activation energy in the forward direction is greater than the activation energy in the reverse direction .
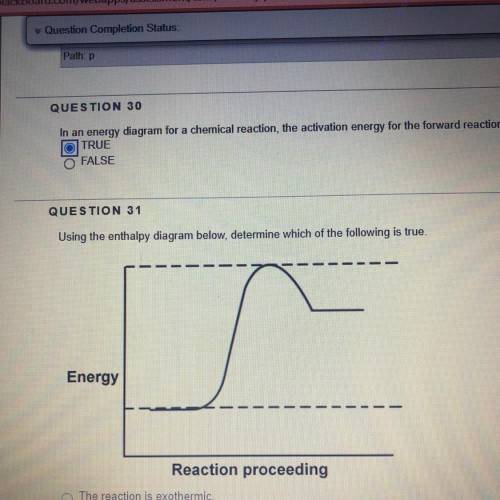

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:40
Darla claims that the first periodic table developed by mendeleev was not completely accurate, so it is not useful at all. harmony argues that it establish the periodic table we use today, making it more credible. who is correct and why? darla is correct, because a model that has any mistakes should be thrown out. darla is correct, because a good model would not need to change. harmony is correct, because mendeleev’s model had all of the information correct in the first version. harmony is correct, because mendeleev’s model made predictions that came true.
Answers: 1


Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1
You know the right answer?
Using the enthalpy diagram below, determine which of the following is true.
1. The reaction is exot...
Questions



Computers and Technology, 03.04.2021 14:03

Mathematics, 03.04.2021 14:03

Mathematics, 03.04.2021 14:03


English, 03.04.2021 14:03

Mathematics, 03.04.2021 14:03



English, 03.04.2021 14:03

Social Studies, 03.04.2021 14:03

Biology, 03.04.2021 14:03


English, 03.04.2021 14:03

Chemistry, 03.04.2021 14:03


English, 03.04.2021 14:03

Business, 03.04.2021 14:03

Mathematics, 03.04.2021 14:03



