
Chemistry, 02.10.2020 19:01 issaaamiaaa15
What error, if any, would the calculated density of the solid have if the material had a hollow center?
1 ) The calculated density would be low, because the volume would be incorrectly measured low.
2) The calculated density would be low, because the volume would be incorrectly measured high.
3)The calculated density would be high, because the volume would be incorrectly measured low.
4)The calculated density would be high, because the volume would be incorrectly measured high.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1


Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1
You know the right answer?
What error, if any, would the calculated density of the solid have if the material had a hollow cent...
Questions


Mathematics, 09.02.2021 20:50

Health, 09.02.2021 20:50



Mathematics, 09.02.2021 20:50


Biology, 09.02.2021 20:50

Mathematics, 09.02.2021 20:50


Mathematics, 09.02.2021 20:50

Advanced Placement (AP), 09.02.2021 20:50

Mathematics, 09.02.2021 20:50

English, 09.02.2021 20:50





Mathematics, 09.02.2021 20:50

English, 09.02.2021 20:50

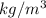 .
.

