

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

You know the right answer?
The atomic weight of a newly discovered element is 10.600 amu. it has two naturally occuring isotope...
Questions


Mathematics, 18.06.2020 01:57


Mathematics, 18.06.2020 01:57

Social Studies, 18.06.2020 01:57



Mathematics, 18.06.2020 01:57









Mathematics, 18.06.2020 01:57

English, 18.06.2020 01:57

Mathematics, 18.06.2020 01:57

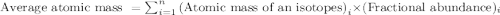
![10.600=[(10.000\times 0.70)+(x\times 0.3)]\\\\x=12](/tpl/images/0316/5804/ea62c.png)


