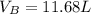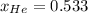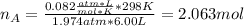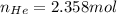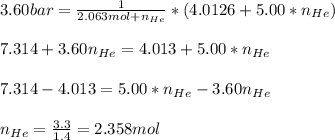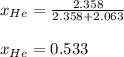Chemistry, 04.10.2020 17:01 devenybates
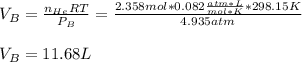
Argon (Ar) and helium (He) are initially in separate compartments of a container at 25°C. The
Ar in compartment A, which has a volume VA of 6.00 L, has a pressure of 2.00 bar. The He in
compartment B of unknown volume V3 has a pressure of 5.00 bar. When the two compartments
are connected and the gases allowed to mix, the total pressure of gas is 3.60 bar. Assume both
gases behave ideally
(a) [4 marks) Determine the volume of compartment B.
(b) [2 marks] Determine the mole fraction of He in the mixture of gases.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
What’s the scientific notation for the number 6,840,000,000
Answers: 1

Chemistry, 22.06.2019 03:00
Explain how the integumentary system plays a crucial role in the ability to maintain homeoestasis
Answers: 1

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2
You know the right answer?
Argon (Ar) and helium (He) are initially in separate compartments of a container at 25°C. The
Ar in...
Questions


Biology, 01.07.2019 19:00

Social Studies, 01.07.2019 19:00

Biology, 01.07.2019 19:00





Biology, 01.07.2019 19:00


Chemistry, 01.07.2019 19:00

Health, 01.07.2019 19:00




History, 01.07.2019 19:00



Mathematics, 01.07.2019 19:00