Chemistry, 07.10.2020 08:01 avadestinyy
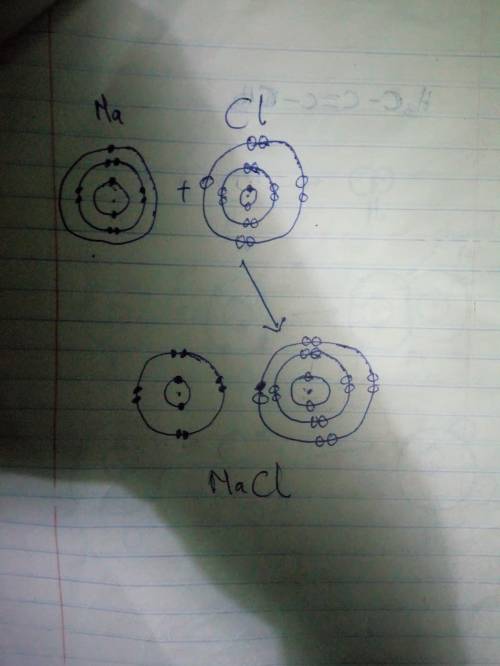
The following figure represents the formation of an ionic compound. Substances A and B are initially uncharged, but when mixed electrons are transferred. Using the figure, identify which substance will form the cation and which will form the anion. Provide a brief (one or two sentences) explanation for your response. (Hint: How does losing electrons affect atomic radii?)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:00
What is the most important factor in determining climates.
Answers: 1

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 23.06.2019 03:10
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2
You know the right answer?
The following figure represents the formation of an ionic compound. Substances A and B are initially...
Questions

Biology, 03.01.2020 13:31

Physics, 03.01.2020 13:31

Mathematics, 03.01.2020 13:31

History, 03.01.2020 13:31

Mathematics, 03.01.2020 13:31


Social Studies, 03.01.2020 13:31








Mathematics, 03.01.2020 13:31

Mathematics, 03.01.2020 13:31

Chemistry, 03.01.2020 13:31

History, 03.01.2020 13:31