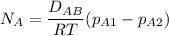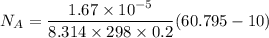The gas CO2 is diffusing at steady state through a tube 0.20 meters long. The tube has a diameter of 0.01 meters and also contains N2 at 298 K. The total pressure inside the tube is constant at 101.32 kPa. The partial pressure of CO2 is 456 mm Hg at one end and 76 mm Hg at the other end. The diffusion coefficient of CO2 in N2 is 1.67 x 10-5 m2 /sec at 298 K. Calculate the molar flux of CO2 in SI units, assuming equimolar counter-diffusion between the CO2 and N2 gases.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1

Chemistry, 23.06.2019 01:00
Which elements are found in glucose, the product of photosynthesis? a. carbon, hydrogen, and oxygen b. carbon and hydrogen c. carbon, nitrogen, and oxygen d. hydrogen, nitrogen, and carbon
Answers: 2

Chemistry, 23.06.2019 03:30
Name 3 types of energy you see being used as you look around a classroom
Answers: 1
You know the right answer?
The gas CO2 is diffusing at steady state through a tube 0.20 meters long. The tube has a diameter of...
Questions

Social Studies, 25.08.2021 16:40

History, 25.08.2021 16:40



Social Studies, 25.08.2021 16:40

Biology, 25.08.2021 16:40


English, 25.08.2021 16:40



Physics, 25.08.2021 16:40


Mathematics, 25.08.2021 16:40

English, 25.08.2021 16:40




Mathematics, 25.08.2021 16:50

Social Studies, 25.08.2021 16:50

Mathematics, 25.08.2021 16:50

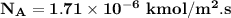
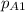 is 456 mm Hg at one end
is 456 mm Hg at one end 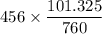
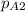 at the other end is 76 mm Hg
at the other end is 76 mm Hg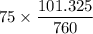
 10 kPa
10 kPa in N
in N