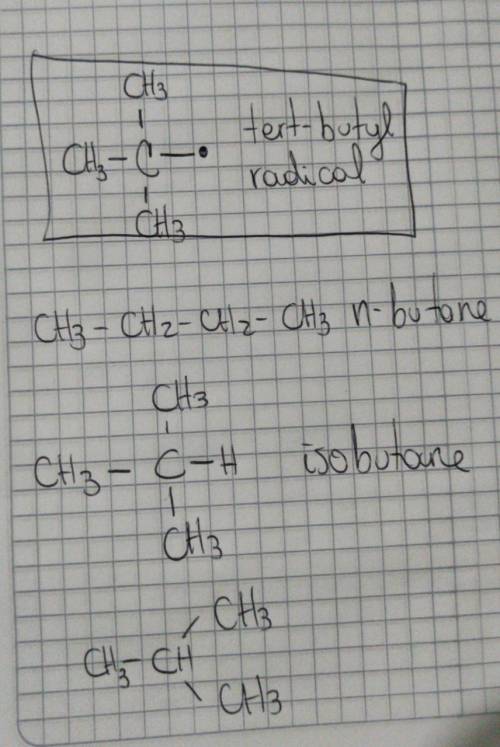
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG (Liquefied Petroleum Gas) cylinders (a common source of cooking gas). It has two arrangements of carbon atoms: a straight chain and a branched chain. Using this information, draw the structure of the tertiary butyl radical that will form upon removal of a hydrogen atom. Draw the molecule on the canvas by choosing buttons from the Tools (for bonds).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2


Chemistry, 23.06.2019 04:20
Which activity describes an application of topographic maps? check all that apply. recreation, such as camping and hiking engineering, such as the construction of roads and buildings science, such as mapping stars in the sky business, such as analyzing population centers science, such as analyzing surface features
Answers: 1
You know the right answer?
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG...
Questions



Mathematics, 20.10.2020 02:01

Mathematics, 20.10.2020 02:01

Computers and Technology, 20.10.2020 02:01


History, 20.10.2020 02:01




Mathematics, 20.10.2020 02:01

History, 20.10.2020 02:01



Biology, 20.10.2020 02:01




Business, 20.10.2020 02:01