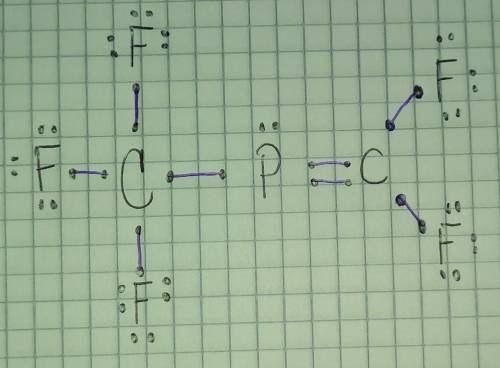Chemistry, 13.10.2020 20:01 ashanti0411
Draw the Lewis structure for CF3PCF2 where all fluorines are bonded to a C atom

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Activity two: just lemons, inc. production here's a one-batch sample of just lemons lemonade production. determine the percent yield and amount of leftover ingredients for lemonade production and place your answers in the data chart. hint: complete stoichiometry calculations for each ingredient to determine the theoretical yield. complete a limiting reactant-to-excess reactant calculation for both excess ingredients. water sugar lemon juice lemonade percent yield leftover ingredients 946.36 g 196.86 g 193.37 g 2050.25 g just lemons lemonade recipe equation: 2 water + sugar + lemon juice = 4 lemonade mole conversion factors: 1 mole of water = 1 cup = 236.59 g 1 mole of sugar = 1 cup = 225 g 1 mole of lemon juice = 1 cup = 257.83 g 1 mole of lemonade = 1 cup = 719.42 g
Answers: 2

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1

Chemistry, 21.06.2019 21:00
Write the chemical symbols for three different atoms or atomic cations with 27 electrons. asap!
Answers: 2

Chemistry, 21.06.2019 22:30
The diagram shows the structures of horse and cat forelimbs. what does the diagram suggest about the evolutionary relationship between these two mammals? a. they have homologous structures, indicating a common ancestor. b. they have analogous structures, indicating a common ancestor. c. they have homologous structures, indicating that they do not have a common ancestor. d. they have analogous structures, indicating that they do not have a common ancestor.
Answers: 2
You know the right answer?
Draw the Lewis structure for CF3PCF2 where all fluorines are bonded to a C atom...
Questions

Geography, 23.08.2019 17:00

English, 23.08.2019 17:00


Biology, 23.08.2019 17:00




History, 23.08.2019 17:00



Biology, 23.08.2019 17:00

Mathematics, 23.08.2019 17:00

English, 23.08.2019 17:00

Chemistry, 23.08.2019 17:00

History, 23.08.2019 17:00

Mathematics, 23.08.2019 17:00

Physics, 23.08.2019 17:00