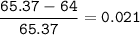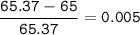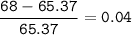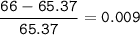Zinc has an average atomic mass of 65.37 amu. Jack is trying to figure out what the most abundant isotope of zinc is, but he doesn't have access to the internet, so the average atomic mass is the only information he has. Jack decides to made an educated guess as to the most abundant isotope of zinc by assuming there are only two isotopes of zinc. If Jack assumes there are only two isotopes of Zn, what he be most likely to decide is zinc's most abundant isotope?
Question 5 options:
Zinc-64
Zinc-65
Zinc-68
Zinc-66

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2



Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
Zinc has an average atomic mass of 65.37 amu. Jack is trying to figure out what the most abundant is...
Questions



Mathematics, 03.07.2019 05:30

Chemistry, 03.07.2019 05:30

Chemistry, 03.07.2019 05:30

Chemistry, 03.07.2019 05:30





Chemistry, 03.07.2019 05:30

Chemistry, 03.07.2019 05:30




Physics, 03.07.2019 05:30

Mathematics, 03.07.2019 05:30

World Languages, 03.07.2019 05:30

World Languages, 03.07.2019 05:30