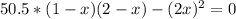Chemistry, 16.10.2020 14:01 williamsjako
A 1.00-L flask is filled with 1.00 moles of H2 and 2.00 moles of I2. The value of the equilibrium constant for the reaction of hydrogen and iodine reacting to form hydrogen iodide is 50.5 under the given conditions. What are the equilibrium concentrations of H2 , I2 , and HI in moles/L? H2 (g) + I2 (g) ⇌ 2HI(g)

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 23.06.2019 08:30
Of the following elements, which is the least reactive? a. c b. h c. li d. he
Answers: 1
You know the right answer?
A 1.00-L flask is filled with 1.00 moles of H2 and 2.00 moles of I2. The value of the equilibrium co...
Questions

Social Studies, 16.01.2021 07:10

History, 16.01.2021 07:10

Mathematics, 16.01.2021 07:10


Social Studies, 16.01.2021 07:10


Mathematics, 16.01.2021 07:10

Mathematics, 16.01.2021 07:10



English, 16.01.2021 07:10


Mathematics, 16.01.2021 07:10

Physics, 16.01.2021 07:10


Mathematics, 16.01.2021 07:10

Mathematics, 16.01.2021 07:10


Mathematics, 16.01.2021 07:10

Mathematics, 16.01.2021 07:10

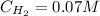
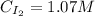
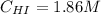
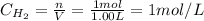
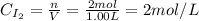
![Kc = \frac{[HI]^{2}}{[H_{2}][I_{2}]} = \frac{(2x)^{2}}{(1-x)(2-x)}](/tpl/images/0811/0470/b43fa.png)