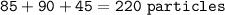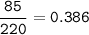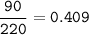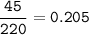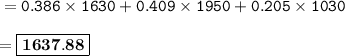Chemistry, 17.10.2020 01:01 nommies005
Calculate the average atomic mass of the mystery element given the isotopic data below.
Sample size: Isotope #1 85 - particles, Isotope #2 - 90 particles, Isotope #3 - 45 particles
Masses: Isotope #1 total mass = 1630 amu, Isotope 2 total mass = 1950 amu, Isotope #3 total mass = 1030 amu

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Is this a scientific model? use complete sentences to explain why or why not. a graphic organizer showing the water cycle
Answers: 3


Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2

Chemistry, 23.06.2019 13:30
The zinc within a copper-plated penny dissolves in hydrochloric acid if the copper coating is filed down in several spots (so that the hydrochloric acid can reach the zinc). the reaction between the acid and the zinc 2h+(aq)+zn(s)→h2(g)+zn2+(aq) . when the zinc in a certain penny dissolves, the total volume of gas collected over water at 25 °c is 0.947 l at a total pressure of 743 mmhg . (vapor pressure of water is 23.78 mmhg at 25 °c .) what mass of hydrogen gas is collected? answer in appropriate significant figures
Answers: 3
You know the right answer?
Calculate the average atomic mass of the mystery element given the isotopic data below.
Sample size...
Questions




English, 27.05.2020 02:03




Chemistry, 27.05.2020 02:03


Computers and Technology, 27.05.2020 02:03

Mathematics, 27.05.2020 02:03


Social Studies, 27.05.2020 02:03

Mathematics, 27.05.2020 02:03