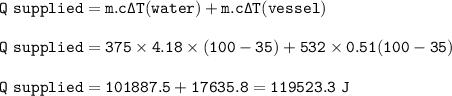Calculate the heat that must be supplied to
a 532 , stainless steel vessel containing 375 g
o...

Chemistry, 18.10.2020 14:01 iziyahh902
Calculate the heat that must be supplied to
a 532 , stainless steel vessel containing 375 g
of water to raise its temperature from 35 C
to the boiling point of water 100°C. Cs. =0.51 J/g. degrees C
Answer in units of J.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3
You know the right answer?
Questions

History, 04.02.2020 09:53

Mathematics, 04.02.2020 09:53

Mathematics, 04.02.2020 09:53


Mathematics, 04.02.2020 09:53


Health, 04.02.2020 09:53

Physics, 04.02.2020 09:53


Business, 04.02.2020 09:53




History, 04.02.2020 09:53


English, 04.02.2020 09:53

History, 04.02.2020 09:53

Mathematics, 04.02.2020 09:53