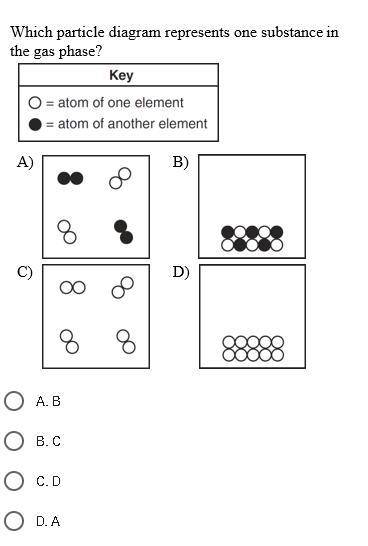
Answer question in picture please.
...

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 06:50
The student repeated the experiment using a higher concentration of acid. the same volume of acid and the same mass of magnesium ribbon were used. what volume of hydrogen gas would have been produced after 60 seconds?
Answers: 1

Chemistry, 23.06.2019 08:30
Which can be observed only in a microscopic view? a) structure of a muscle cell b) shape of a soybean plant c) foam insulation d) x-ray of a knee joint
Answers: 2
You know the right answer?
Questions






English, 05.01.2021 20:20

Mathematics, 05.01.2021 20:20

History, 05.01.2021 20:20

Advanced Placement (AP), 05.01.2021 20:20

Social Studies, 05.01.2021 20:20

Chemistry, 05.01.2021 20:20

World Languages, 05.01.2021 20:20


Mathematics, 05.01.2021 20:20

Mathematics, 05.01.2021 20:20

Mathematics, 05.01.2021 20:20


English, 05.01.2021 20:20

Mathematics, 05.01.2021 20:20

Mathematics, 05.01.2021 20:20