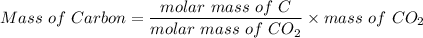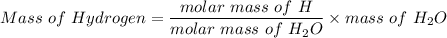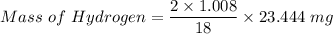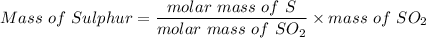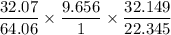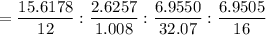Chemistry, 21.10.2020 16:01 PONBallfordM89
A 32.149 mg sample of a chemical known to contain only carbon hydrogen sulfur and oxygen is put into a combustion analysis apparatus yielding 57.271 mg of co2 and 23.444 mg of h2o. In another experiment 22.345 mg of the compound is reacted with excess oxygen to produce 9.656 mg of sulfur dioxide

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

Chemistry, 22.06.2019 20:00
Listenbase your answer to the question on the information below.nuclear radiation is harmful to living cells, particularly to fast-growing cells, such as cancer cells and blood cells. an external beam of the radiation emitted from a radioisotope can be directed on a small area of a person to destroy cancer cells within the body.cobalt-60 is an artificially produced radioisotope that emits gamma rays and beta particles. one hospital keeps a 100.0-gram sample of cobalt-60 in an appropriate, secure storage container for future cancer treatment.which choice represents the correct product for the beta decay of the co-60? fe-60ni-60fe-61ni-61
Answers: 2

You know the right answer?
A 32.149 mg sample of a chemical known to contain only carbon hydrogen sulfur and oxygen is put into...
Questions

English, 18.03.2021 22:50







Mathematics, 18.03.2021 22:50


Mathematics, 18.03.2021 22:50

Mathematics, 18.03.2021 22:50





Mathematics, 18.03.2021 22:50



English, 18.03.2021 22:50