
Chemistry, 21.10.2020 16:01 ilovecatsomuchlolol
An aqueous solution of Pb(NO3)2 is made by placing 275 g of solid Pb(NO3)2 into a volumetric flask and adding water to the 1.00 L mark (assume that 775g of water has been added to achieve this total solution volume). (Assume MW of Pb(NO3)2 = 331g/mole) A) What is the molarity (M) of this solution? B) What is the molality (m) of this solution? C) What is the mass % of Pb(NO3)2 in this solution? D) What is the mole fraction of Pb(NO3)2 present in this solution?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Si una estrella no tiene paralaje medible, ¿qué puedes inferir?
Answers: 1


Chemistry, 22.06.2019 06:40
Which statement is usually true about the relationship between activation energy and reaction rates? low activation energy barriers result in low rates. high activation energy barriers result in low rates. low activation energy barriers result in no reaction. high activation energy barriers result in no reaction.
Answers: 3

You know the right answer?
An aqueous solution of Pb(NO3)2 is made by placing 275 g of solid Pb(NO3)2 into a volumetric flask a...
Questions

Mathematics, 06.05.2020 02:42

Biology, 06.05.2020 02:42



Chemistry, 06.05.2020 02:42








Mathematics, 06.05.2020 02:42







Mathematics, 06.05.2020 02:42

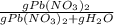 *100%
*100% 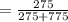 * 100% = 26.2%
* 100% = 26.2%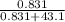 = 0.0189
= 0.0189


