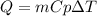Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1
You know the right answer?
You have a 50.0g samples of silver, a 50g sample of iron, and a 50g sample of water. You add 100 J o...
Questions

Biology, 24.07.2019 06:20


Chemistry, 24.07.2019 06:20


Biology, 24.07.2019 06:20

Mathematics, 24.07.2019 06:20

Chemistry, 24.07.2019 06:20

Business, 24.07.2019 06:20



Social Studies, 24.07.2019 06:20



Social Studies, 24.07.2019 06:20




History, 24.07.2019 06:20

Chemistry, 24.07.2019 06:20

Business, 24.07.2019 06:20