
Chemistry, 22.10.2020 19:01 pancakefox7
Suppose you are studying the kinetics of the iodine-catalyzed decomposition of hydrogen peroxide. 2 H 2 O 2 ⟶ 2 H 2 O + O 2 If you determine the initial rate is 7.50 × 10 − 4 M/s when [ H 2 O 2 ] = 0.546 M and [ K I ] = 0.212 M , what is the rate constant? Assume that the order of both reactants is 1.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2


Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2
You know the right answer?
Suppose you are studying the kinetics of the iodine-catalyzed decomposition of hydrogen peroxide. 2...
Questions



History, 23.06.2019 06:30

Social Studies, 23.06.2019 06:30



History, 23.06.2019 06:30

Chemistry, 23.06.2019 06:30

Chemistry, 23.06.2019 06:30


History, 23.06.2019 06:30



Biology, 23.06.2019 06:30


Biology, 23.06.2019 06:30

History, 23.06.2019 06:30

History, 23.06.2019 06:30


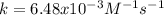
![r=k[H_2O_2][KI]](/tpl/images/0832/1312/1c29a.png)
![k=\frac{r}{[H_2O_2][KI]}=\frac{7.50x10^{-4}M/s}{0.546M*0.212M}\\ \\k=6.48x10^{-3}M^{-1}s^{-1}](/tpl/images/0832/1312/953ba.png)


